The first full statements of the law came in 1850 from Rudolf Clausius, and from William Rankine. In 1842, Julius Robert von Mayer made a statement that was expressed by Clifford Truesdell (1980) in the rendition "in a process at constant pressure, the heat used to produce expansion is universally interconvertible with work", but this is not a general statement of the first law. 
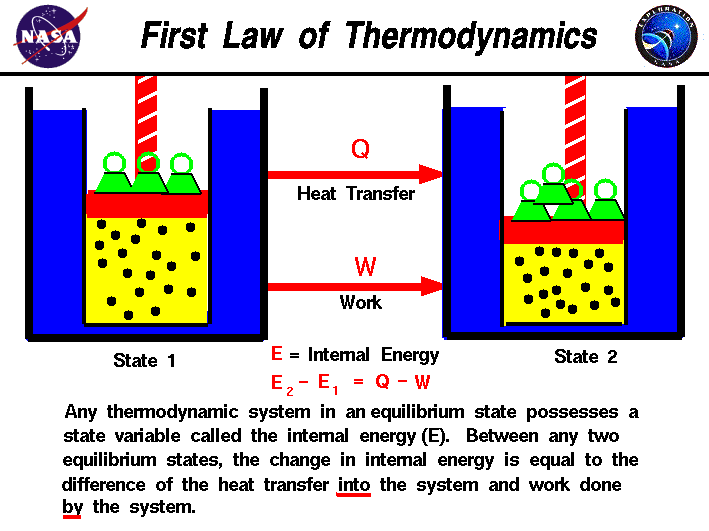
This law was later recognized as a consequence of the first law of thermodynamics, but Hess's statement was not explicitly concerned with the relation between energy exchanges by heat and work. In 1840, Germain Hess stated a conservation law ( Hess's Law) for the heat of reaction during chemical transformations. Empirical developments of the early ideas, in the century following, wrestled with contravening concepts such as the caloric theory of heat. In the first half of the eighteenth century, French philosopher and mathematician Émilie du Châtelet made notable contributions to the emerging theoretical framework of energy by proposing a form of the law of conservation of energy that recognized the inclusion of kinetic energy. For such considerations, thermodynamics also defines the concept of open systems, closed systems, and other types. Many systems in practical applications require the consideration of internal chemical or nuclear reactions, as well as transfers of matter into or out of the system. The ideal isolated system, of which the entire universe is an example, is often only used as a model. Δ U = Q − W, is recovered) to make the system work continuously. The first law for a thermodynamic process is often formulated as To take relativity into account, any reference frame difference, such as relative motion, needs to be accounted for.) (This is in the context of a ' rest frame', a system observed from the same frame of reference. Energy can be transformed from one form to another, but can be neither created nor destroyed.īy the mass-energy equivalence principle in Einstein's famous E = m c 2 equation, matter and energy can be converted into one another, without violating the First Law. The law of conservation of energy states that the total energy of any isolated system, which cannot exchange energy or matter, is constant. The law also defines the internal energy of a system, an extensive property for taking account of the balance of energies in the system. It distinguishes in principle two forms of energy transfer: heat, and thermodynamic work, for a system of a constant amount of matter. While there are many subtleties and implications that may be more precisely captured in more complex formulations, this is the essential principle of the First Law. A simple formulation is: "The total energy in a system remains constant, although it may be converted from one form to another." Another common phrasing is that "energy can neither be created nor destroyed" (in a "closed system"). 
The first law of thermodynamics is a formulation of the law of conservation of energy, adapted for thermodynamic processes.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
